Gelatinous White Ppt . (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. The above statement can be represented as a reaction and is given as follows: they look very different. Dissolve a small quantity of the substance in water. double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. Place about 5cm 3 of the solution into a test tube. Add a few drops of sodium hydroxide solution. Ammonium + turns red litmus blue. Faint white unless very low conc. Which dissolves in excess of naoh. The correct option is b.
from www.slideserve.com
double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. Add a few drops of sodium hydroxide solution. Faint white unless very low conc. (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. The above statement can be represented as a reaction and is given as follows: Ammonium + turns red litmus blue. Place about 5cm 3 of the solution into a test tube. The correct option is b. (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. Dissolve a small quantity of the substance in water.
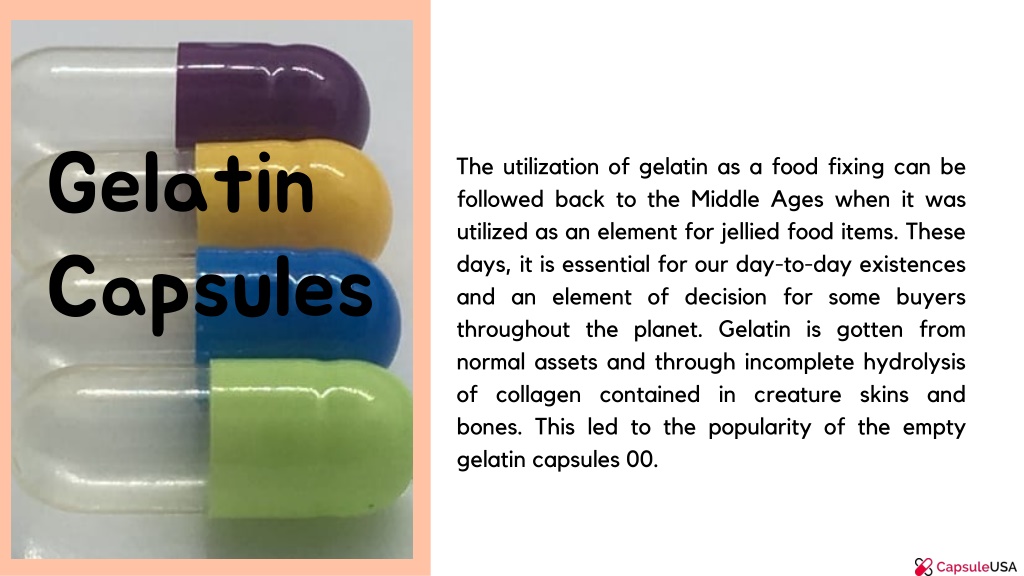
PPT Key Features of Gelatin Capsules PowerPoint Presentation, free
Gelatinous White Ppt Ammonium + turns red litmus blue. (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. The above statement can be represented as a reaction and is given as follows: (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. Add a few drops of sodium hydroxide solution. double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. Which dissolves in excess of naoh. they look very different. Dissolve a small quantity of the substance in water. Ammonium + turns red litmus blue. Faint white unless very low conc. Place about 5cm 3 of the solution into a test tube. The correct option is b.
From dxovatzox.blob.core.windows.net
Zinc Hydroxide Color at Thelma Collins blog Gelatinous White Ppt (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. Place about 5cm 3 of the solution into a test tube. Faint white unless very low conc. Add a few drops of. Gelatinous White Ppt.
From www.youtube.com
Gelatin — Properties, Functions and Food Applications YouTube Gelatinous White Ppt Add a few drops of sodium hydroxide solution. Place about 5cm 3 of the solution into a test tube. Faint white unless very low conc. The above statement can be represented as a reaction and is given as follows: Which dissolves in excess of naoh. they look very different. (iii) when naoh is added to the given soln.,. Gelatinous White Ppt.
From www.slideserve.com
PPT Manufacture of Gelatin PowerPoint Presentation, free download Gelatinous White Ppt The above statement can be represented as a reaction and is given as follows: (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. Add a few drops of sodium hydroxide solution. The correct option is b. Ammonium + turns red litmus blue. Faint white unless very low conc. Which dissolves in. Gelatinous White Ppt.
From www.slideserve.com
PPT GELATIN LIQUEFACTION TEST PowerPoint Presentation, free download Gelatinous White Ppt The above statement can be represented as a reaction and is given as follows: (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. Place about 5cm 3 of the solution into a test tube. double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. Ammonium + turns. Gelatinous White Ppt.
From www.slideserve.com
PPT Zooplankton PowerPoint Presentation, free download ID6020879 Gelatinous White Ppt Ammonium + turns red litmus blue. The above statement can be represented as a reaction and is given as follows: (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. they look very different. The correct option is b. Add a few drops of sodium hydroxide solution. double. Gelatinous White Ppt.
From www.dreamstime.com
Gelatin stock image. Image of granules, collagen, white 44438203 Gelatinous White Ppt (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. Dissolve a small quantity of the substance in water. they look very different. The correct option is b. Ammonium + turns red litmus blue. Place about 5cm 3 of the solution into a test tube. (ii) gelatinous white. Gelatinous White Ppt.
From www.slideserve.com
PPT Autotrophs PowerPoint Presentation, free download ID8684464 Gelatinous White Ppt The above statement can be represented as a reaction and is given as follows: Ammonium + turns red litmus blue. Dissolve a small quantity of the substance in water. (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. Place about 5cm 3 of the solution into a test tube.. Gelatinous White Ppt.
From www.researchgate.net
Intrasurgical image of case 4 showing a white gelatinous material Gelatinous White Ppt (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. Ammonium + turns red litmus blue. they look very different. Faint white unless very low conc. Place about 5cm 3 of the solution into a test tube. Dissolve a small quantity of the substance in water. The above statement can be. Gelatinous White Ppt.
From www.slideserve.com
PPT Clostridium PowerPoint Presentation, free download ID3031736 Gelatinous White Ppt double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. Place about 5cm 3 of the solution into a test tube. Ammonium + turns red litmus blue. The above statement can be represented as a reaction and is given as follows: Add a few drops of sodium hydroxide solution. (iii) when naoh is added to the given. Gelatinous White Ppt.
From www.reddit.com
Ghostly gelatinous all white fungus with tiny pores underneath, central Gelatinous White Ppt Ammonium + turns red litmus blue. Add a few drops of sodium hydroxide solution. (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. Place about 5cm 3 of the solution into a test tube. double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. The correct option. Gelatinous White Ppt.
From capsuleconnection.com
Whole White Empty Gelatin Capsules Gelatinous White Ppt Place about 5cm 3 of the solution into a test tube. double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. they look very different. Which dissolves in excess of naoh. Add a few drops of sodium hydroxide solution. Dissolve a small quantity of the substance in water. (ii) gelatinous white precipitate of zinc hydroxide is. Gelatinous White Ppt.
From www.numerade.com
SOLVEDPart B Observations lons Limited NaOH Excess NaOH Limited NHz Gelatinous White Ppt they look very different. Add a few drops of sodium hydroxide solution. Which dissolves in excess of naoh. double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. Faint white unless very low conc. (iii) when naoh is. Gelatinous White Ppt.
From www.youtube.com
Aluminiuminsolution Test for Aluminium in Solution and Gelatinous Gelatinous White Ppt Place about 5cm 3 of the solution into a test tube. (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. Which dissolves in excess of naoh. The correct option is b. double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. The above statement can be represented. Gelatinous White Ppt.
From www.slideserve.com
PPT What Is The Difference Between Soft Gelatin And Hard Gelatin Gelatinous White Ppt Ammonium + turns red litmus blue. Add a few drops of sodium hydroxide solution. The correct option is b. they look very different. Place about 5cm 3 of the solution into a test tube. The above statement can be represented as a reaction and is given as follows: (iii) when naoh is added to the given soln., zn(no. Gelatinous White Ppt.
From www.doubtnut.com
A gelatinous white ppt is formed which is soluble in excess of alkali. Gelatinous White Ppt they look very different. Which dissolves in excess of naoh. (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. Faint white unless very low conc. Place about 5cm 3 of the solution into a test tube. Add a. Gelatinous White Ppt.
From thecontentauthority.com
Gelatinization vs Gelatinous Meaning And Differences Gelatinous White Ppt The above statement can be represented as a reaction and is given as follows: The correct option is b. Place about 5cm 3 of the solution into a test tube. Ammonium + turns red litmus blue. Dissolve a small quantity of the substance in water. double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. Which dissolves in. Gelatinous White Ppt.
From www.slideserve.com
PPT Health Benefits of Gelatin Capsules PowerPoint Presentation, free Gelatinous White Ppt (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form a gelatinous white ppt. Faint white unless very low conc. Place about 5cm 3 of the solution into a test tube. they look very different.. Gelatinous White Ppt.
From www.slideserve.com
PPT Gelatin PowerPoint Presentation, free download ID9511590 Gelatinous White Ppt double displacement reactionprecipitation reactionzinc sulphate with sodium hydroxideznso4 with naohgelatinous white. Add a few drops of sodium hydroxide solution. (ii) gelatinous white precipitate of zinc hydroxide is formed which becomes colourless solution on adding excess of. Ammonium + turns red litmus blue. (iii) when naoh is added to the given soln., zn(no 3) 2 reacts to form. Gelatinous White Ppt.